 
The negative individuals were considered previously uninfected.Īll the symptomatic and asymptomatic infections occurred prior to vaccination during the first wave (from the 21st February to the 11th June 2020) and the second wave (from 14th September to 31th December 2020) in Italy. Previous infection was defined as SARS-CoV-2 N-protein IgG positivity before vaccination and/or history of positive polymerase chain reaction result from a nasopharyngeal swab. The positive threshold was 1.4 signal to cutoff-ratio. In April, June and November 2020, before the first dose of BNT162b2/Pfizer vaccine, the antibodies against SARS-CoV-2 nucleocapsid (SARS-CoV-2 N-protein IgG) (Abbott Diagnostics) were measured in all HCWs at the Poliambulanza Foundation Hospital Brescia, a 600-bed tertiary care hospital in Northern Italy. This observational prospective study was designed to verify durability of a protective immune response via antibodies against the viral receptor-binding domain (RBD) spike (S)-protein (RBD S-protein IgG) after the second-dose administration of BNT162b2/Pfizer vaccine among Health Care Workers (HCWs). Other studies also found a modest decline in the frequency of T cells at 6 months and a robust expansion in response to antigen and recognition of spike peptides from the delta variant. Previous studies have found a significant waning of humoral responses within 6 months after receipt of the second dose of BNT162b2 with a continuous decrease in anti-spike IgG titers. However, long-term effectiveness remains undetermined, and there is evidence of waning immunity in terms of antibody dynamics. 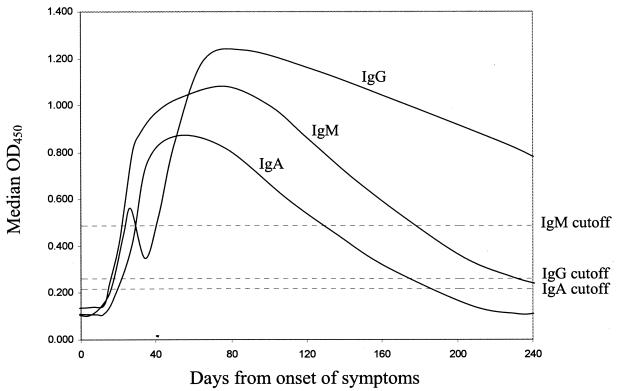
The booster dose of the vaccine could be delayed after the second dose in previously naturally infected subject and it could have an important strategic impact on the organization of the future COVID-19 vaccination campaign.Ī two-dose regimen of the BioNTech/Pfizer mRNA BNT162b2 severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccine was demonstrated to be highly effective in preventing symptomatic COVID-19, both in clinical trials and real-world settings. The limitations of this study prevent us to draw definitive conclusions on the antibody titers and on the role of a previous SARS-CoV-2 infection in influencing the levels of post-vaccine RBD S-protein IgG. Moreover, our data suggest that the risk of a new SARS-CoV-2 infection was higher in the previously uninfected HCWs than in those who had already contracted natural viral infection. 
We report that the mean level of post vaccinal RBD S-protein IgG was significantly higher in the previously infected HCWs than in previously uninfected subjects at 45 and 240 days after the second-dose vaccine. No new infections were found in previously SARS-CoV-2 infected subjects. In addition, in the HCWs group without previous infection we reported 53 new SARS-CoV-2 infections and they had a mean level of RBD S-protein IgG antibodies of 1039 AU/mL (95% CI 157–4237) at 240 days. When comparing the change in mean antibody levels overtime, the previously infected HCWs presented a 72.9% reduction in RBD S-protein IgG while in the previously uninfected HCWs the reduction was 88.0%. Similarly, at 240 days in previously infected subjects the antibody titer was of 8145 AU/mL (95% CI 347–80,000) and significantly higher ( p < 0.001) than that observed in the 1121 previously uninfected HCWs with a mean antibody level of 1271 AU/mL (95% CI 50–80,000). The mean level of the RBD S-protein IgG detected 45 days after the second dose of the vaccine was 30,041 AU/mL (95% CI 145–80,000) for the 250 previously infected HCWs and it was significantly higher ( p < 0.001) than that observed in the 1121 previously uninfected subject with the mean level of 10,604 AU/mL (95% CI 165–62,241). 
The Mann–Whitney U test was used to compare mean levels of RBD S-Protein IgG among previously infected and uninfected HCWs. Previous infection was defined as antibodies against SARS-CoV-2 nucleocapsid positivity (Abbott Diagnostics) before vaccination. In all HCWs at the Poliambulanza Foundation Hospital Brescia (Italy) were quantified the levels of RBD S-Protein IgG (Abbott Diagnostics) at 45 and 240 days after the second-dose vaccine. This observational prospective study was designed to verify durability of antibodies against the viral receptor-binding domain (RBD) spike (S)-protein (RBD S-Protein IgG) after the second-dose administration of the vaccine among Health Care Workers (HCWs). The long-term effectiveness of BNT162b2/Pfizer vaccine remains undetermined. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
